Usage of Caustic soda in the process of extracting aluminum
Aluminum , is the largest caustic soda application segment in the chlor-alkali market. Caustic soda is used in the process of extracting alumina from bauxite ore. It dissolves bauxite ore, which is the raw material for it production. In this process, pure alumina is separated from bauxite ore. The collected alumina is further subjected to calcination.
How alumina reacts with caustic soda?
Therefore, when alumina , which is an amphoteric metal, reacts with caustic soda, sodium aluminate is formed and hydrogen gas is evolved as a result.
The reaction is as follows:
Aluminum + 2 Na O H Caustic soda + 2H2O → 2Na A lO2 Sodium met aluminite + 3H2 ↑ Hydrogen gas
This reaction is very exothermic.
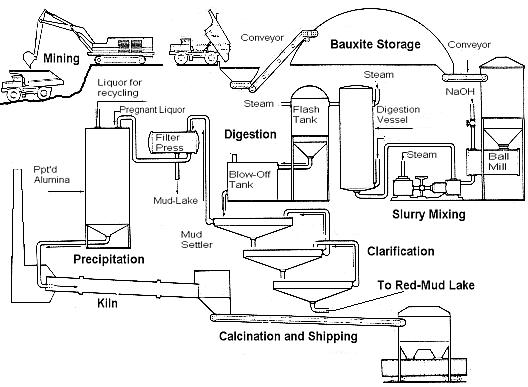
what is Bayer process for product alumina?
The Bayer process is the principal industrial means of refining bauxite to produce alumina (aluminum oxide) and was developed by Carl Josef Bayer. Bauxite, the most important ore of alumina, contains only 30-60% aluminum oxide (Al203), the rest being a mixture of silica, various iron oxides, and titanium dioxide. The alumina oxide must be further purified before it can be refined into alumina metal. For this process used caustic soda, It dissolves bauxite ore.
The position of caustic soda in the category of chlor-alkali products
Chlor-alkali products include chlorine, caustic soda, and soda ash. The main technologies used in the chlor-alkali process are membrane cell, diaphragm cell and mercury cell to produce chlorine and caustic soda. Chlorine and caustic soda are produced from the electrolysis of an aqueous solution of sodium chloride (common salt or Na Cl). Soda ash is produced from the mining and refining of Tron ore (natural) and also through the Solvay process (synthetic). The chlor-alkali process is energy intensive.
Difference between aluminum and alumina?
Aluminum is a chemical element, but alumina is an aluminum containing compound, alumina is a compound which includes aluminum chemical element.